Let’s discuss the question: how many protons does ga3+ have. We summarize all relevant answers in section Q&A of website Linksofstrathaven.com in category: Blog Finance. See more related questions in the comments below.
How many neutrons does ga3 have?
…
How many electrons are in gallium?
| Name | Gallium |
|---|---|
| Number of Neutrons | 39 |
| Number of Electrons | 31 |
How many electrons are present in ga3 +?
Here, the electron configuration of gallium ion(Ga3+) is 1s2 2s2 2p6 3s2 3p6 3d10. This electron configuration shows that gallium ion(Ga3+) has three shells and the last shell has eighteen electrons. Since the last shell of a gallium ion has eighteen electrons, the valence electrons of gallium ions(Ga3+) are eighteen.
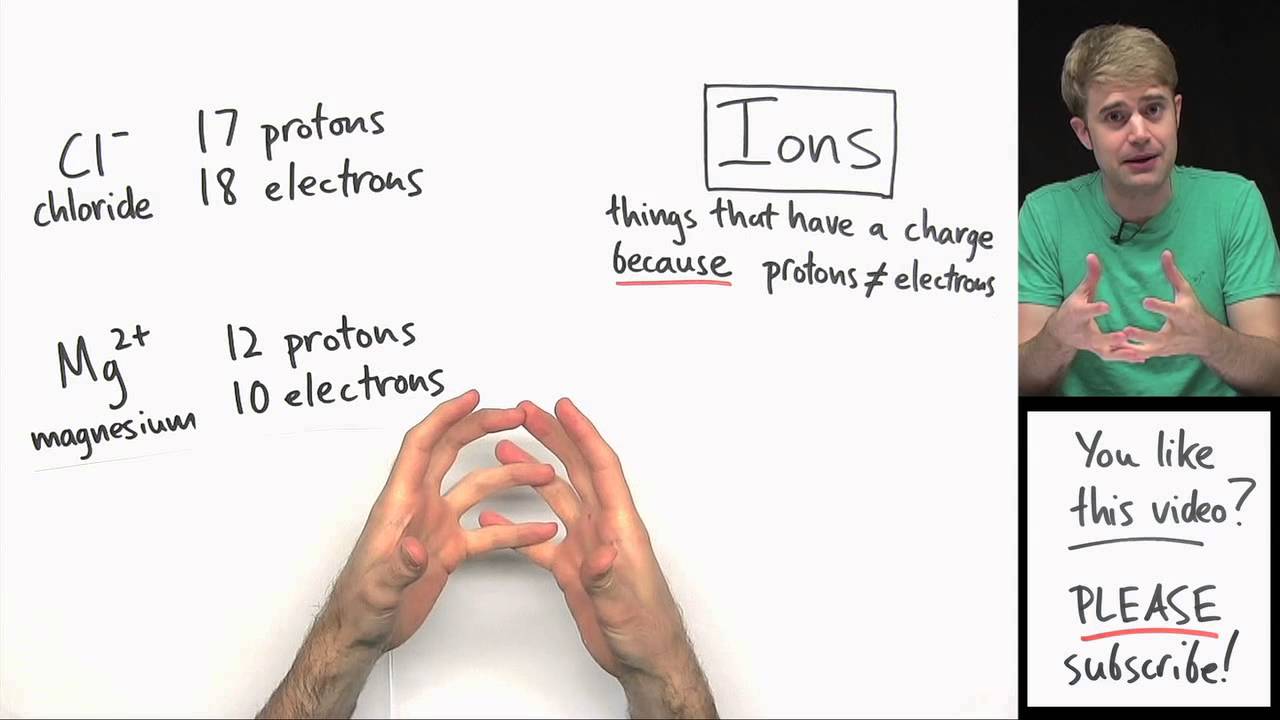
Huge Misconception: Protons, Electrons, Atoms, and Ions
Images related to the topicHuge Misconception: Protons, Electrons, Atoms, and Ions

How many protons does gallium have?
What is the atomic number for Ga 3+?
…
4.3Related Element.
| Element Name | Gallium |
|---|---|
| Atomic Number | 31 |
How many protons are in Ga 3?
Gallium, Ga has 31 protons and 31 electrons.
How many protons and electrons does gallium have?
Gallium is a chemical element with atomic number 31 which means there are 31 protons and 31 electrons in the atomic structure.
How many orbitals does gallium have?
| Atomic Number | 31 |
|---|---|
| Number of Neutrons | 39 |
| Shell structure (Electrons per energy level) | [2, 8, 18, 3] |
| Electron Configuration | [Ar] 3d10 4s2 4p1 |
| Valence Electrons | 4s2 4p1 |
What is the orbital notation for Ga?
Gallium(Ga) excited state electron configuration
Ground state electron configuration of gallium is 1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p1. In the gallium ground-state electron configuration, an electron of the 4p orbital is located in the 4px sub-orbitals. The p-orbital has three sub-orbitals.
Which element is 1s22s22p63s23p3?
The 1s orbital
Given that the electron configuration for phosphorus is 1s22s22p63s23p3, answer the following questions: a.
How many protons does in have?
…
2.1 Electrons, Protons, Neutrons, and Atoms.
| Elementary Particle | Charge | Mass |
|---|---|---|
| Proton | +1 | 1 |
| Neutron | 0 | 1 |
| Electron | −1 | ~0 |
How To Calculate The Number of Protons, Neutrons, and Electrons – Chemistry
Images related to the topicHow To Calculate The Number of Protons, Neutrons, and Electrons – Chemistry
How many protons are in Boron?
How many protons does Gallium 70?
Diagram of the nuclear composition, electron configuration, chemical data, and valence orbitals of an atom of gallium-70 (atomic number: 31), an isotope of this element. The nucleus consists of 31 protons (red) and 39 neutrons (orange).
What element is Ga?
gallium (Ga), chemical element, metal of main Group 13 (IIIa, or boron group) of the periodic table. It liquefies just above room temperature.
What is gallium worth?
| Products | Price Range | Date |
|---|---|---|
| Germanium Dioxide | 5,700~5,900 | Apr 21, 2022 |
| Gallium | 2,580~2,630 | Apr 21, 2022 |
| High Purity Ga(6N) | 2,780~2,830 | Apr 21, 2022 |
| High Purity Ga(7N) | 3,050~3,100 | Apr 21, 2022 |
What is GaN in chemistry?
Gallium nitride (GaN) is a very hard, mechanically stable wide bandgap semiconductor. With higher breakdown strength, faster switching speed, higher thermal conductivity and lower on-resistance, power devices based on GaN significantly outperform silicon-based devices.
How many neutrons does gallium +3 have?
The nucleus consists of 31 protons (red) and 39 neutrons (orange). 31 electrons (white) successively occupy available electron shells (rings). The stability of an element’s outer (valence) electrons determines its chemical and physical properties.
How many protons does Gallium 71 have?
| Properties of Gallium-71 Isotope: | GALLIUM-71 |
|---|---|
| Nucleon Number (A) | 71 |
| Proton Number (Z) | 31 |
| Half-life | Stable |
| Spin | 1.5 |
How do you find the protons neutrons and electrons?
- number of protons = atomic number.
- number of electrons = atomic number.
- number of neutrons = mass number – atomic number.
How many electrons does gallium have?
Gallium atoms have 31 electrons and the shell structure is 2.8. 18.3. The ground state electron configuration of ground state gaseous neutral gallium is [Ar].
How Many Protons, Neutrons, And Electrons Does The Atom 39-K Have?
Images related to the topicHow Many Protons, Neutrons, And Electrons Does The Atom 39-K Have?

How many protons does the only liquid metal have?
Mercury atoms have 80 electrons and 80 protons with 122 neutrons in the most abundant isotope. Under standard conditions mercury is a shiny, heavy, silvery liquid. It is the only metal that is liquid at room temperature. It will evaporate into the air at room temperature.
What is C on the periodic table?
Carbon – Element information, properties and uses | Periodic Table.
Related searches
- how many protons and electrons in p3-
- how many neutrons does ga3 have
- how many electrons does ga have
- li protons and electrons
- how many protons does p3- have
- how many protons can the third shell hold
- ga3+ neutrons
- ga3 neutrons
- how many neutrons does ga3+ have
- ga3+ atomic number
- how many electrons does cl have
- ga3 atomic number
- determine the number of protons and neutrons in the isotope fe 58
- how many terms of gp 3 3^2
- how many protons does n-3 have
- how many protons does ga3+ have
- how many protons does helium 3 have
- how many protons are in an atom of ga
- ga protons neutrons electrons
- how many electrons does cl- have
- how many protons does al+3 have
- how many protons in p3-
Information related to the topic how many protons does ga3+ have
Here are the search results of the thread how many protons does ga3+ have from Bing. You can read more if you want.
You have just come across an article on the topic how many protons does ga3+ have. If you found this article useful, please share it. Thank you very much.