Let’s discuss the question: how many neutrons does an atom of tungsten have. We summarize all relevant answers in section Q&A of website Linksofstrathaven.com in category: Blog Finance. See more related questions in the comments below.
How many neutrons does tungsten contain?
Tungsten’s atomic mass is 183. The atomic mass is obtained by calculating the average number of neutrons plus the protons in the tungsten isotope. There are five different stable isotopes of tungsten containing 108, 110, 106, 109, and 112 neutrons. The average of these isotopes is 109.
How many neutrons does an atom of tungsten have Brainly?
tungsten is an element with 74 protons and 109 neutrons .
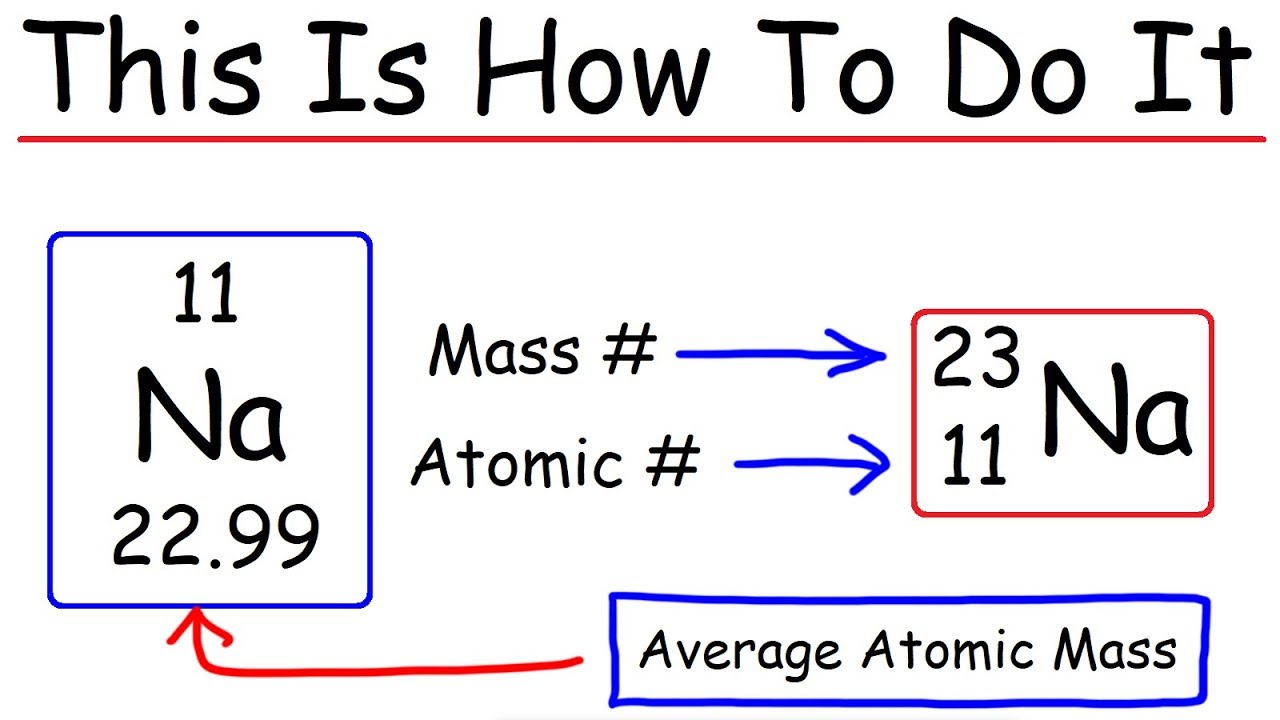
How To Calculate The Number of Protons, Neutrons, and Electrons – Chemistry
Images related to the topicHow To Calculate The Number of Protons, Neutrons, and Electrons – Chemistry

How many electrons and neutrons does tungsten have?
| Atomic Number | 74 |
|---|---|
| Number of Electrons (with no charge) | 74 |
| Number of Protons | 74 |
| Mass Number | 184 |
| Number of Neutrons | 110 |
How many atoms are there in tungsten?
| atomic number | 74 |
|---|---|
| atomic weight | 183.85 |
| melting point | 3,410 °C (6,152 °F) |
| boiling point | 5,660 °C (10,220 °F) |
| density | 19.3 grams/cm3 at 20 °C (68 °F) |
What is tungsten atom?
Tungsten is a metal in group VIB of the periodic table with atomic number 74, an atomic weight of 183.85, and a density of 19.3 Mg/m3. Its melting point is 3410 C, and it boils at 5930 C. The electronic configuration of Tungsten is (Xe)(4f14)(5d4)(6s2).
How many protons neutrons and electrons does tungsten have?
The atomic number of tungsten is 74, so a tungsten atom has 74 protons. Mass number = number of protons + number of neutrons= 184. So number of neutrons = relative atomic mass – number of protons = 184 – 74 = 110. Number of electrons = number of protons.
How many neutrons does an atom of tungsten have round your answer to the nearest whole number?
Thus the number of neutrons in Tungsten atom, rounded to the nearest whole number is 110.
What is tungsten atomic number how many electrons does tungsten have?
Tungsten has 74 electrons. This is easy to determine if you look at the Periodic Table of Elements. The atomic number of tungsten is 74.
What is the atomic weight of tungsten?
How many neutrons does tungsten 184 have?
| Properties of Tungsten-184 Isotope: | TUNGSTEN-184 |
|---|---|
| Neutron Number (N) | 110 |
| Atomic Number (Z) | 74 |
| Mass Number (A) | 184 |
| Nucleon Number (A) | 184 |
How many electrons does tungsten +4 have?
Tungsten atoms have 74 electrons and the shell structure is 2.8.
How many shells are in tungsten?
| Classification: | Tungsten is a transition metal |
|---|---|
| Protons: | 74 |
| Neutrons in most abundant isotope: | 110 |
| Electron shells: | 2,8,18,32,12,2 |
| Electron configuration: | [Xe] 4f14 5d4 6s2 |
Warning: DO NOT TRY—Seeing How Close I Can Get To a Drop of Neutrons
Images related to the topicWarning: DO NOT TRY—Seeing How Close I Can Get To a Drop of Neutrons

How many neutrons does boron have?
Boron is a nonmetallic element, which has five protons in its nucleus along with five to six neutrons, resulting in two stable isotope forms: 10B (abundance of 19.8%) and 11B (abundance of 80.2%), averaging its atomic weight at 10.81Da [1].
What’s the electron configuration for tungsten?
How dense is tungsten?
With a density of 19.28 g/cm³, tungsten is also among the heaviest metals which is why it is used among other things for specific mass balancing.
How many protons are in tungsten?
How is tungsten made?
It’s made by “cementing” very hard tungsten monocarbide (WC) grains in a binder matrix of tough cobalt metal by liquid phase sintering. The result changed the history of tungsten: a material which combines high strength, toughness and high hardness.
What is tungsten class 8?
Tungsten is a solid grayish-white metal that is highly malleable and is often joined with other metals as alloys to increase durability and hardness.
How many neutrons does tungsten 183 have?
| Properties of Tungsten-183 Isotope: | TUNGSTEN-183 |
|---|---|
| Neutron Number (N) | 109 |
| Atomic Number (Z) | 74 |
| Mass Number (A) | 183 |
| Nucleon Number (A) | 183 |
What group is tungsten in?
| Group | 6 | 3414°C, 6177°F, 3687 K |
|---|---|---|
| Atomic number | 74 | 183.84 |
| State at 20°C | Solid | 182W, 184W, 186W |
| Electron configuration | Xe] 4f145d46s2 | 7440-33-7 |
| ChemSpider ID | 22403 | ChemSpider is a free chemical structure database |
How many neutrons are present in the nucleus of a phosphorus atom?
We know that the atomic number of phosphorus is 15, therefore the number of protons present is also 15. The number of neutrons present in phosphorus atom= 31 – 15 = 16. Therefore, the number of neutrons present in the phosphorus atom is 16.
How do you find the neutrons of tungsten?
…
Chemistry Examples.
| 74 | Atomic Number |
|---|---|
| Tungsten | Element Name |
| 183.84 | Average Atomic Mass |
Calculating the Protons, Neutrons and Electrons for an Atom
Images related to the topicCalculating the Protons, Neutrons and Electrons for an Atom

What is the total number of protons of palladium atom?
Protons and Neutrons in Palladium
Palladium is a chemical element with atomic number 46 which means there are 46 protons in its nucleus. Total number of protons in the nucleus is called the atomic number of the atom and is given the symbol Z.
What element has 131 atomic weight and with 77 neutrons?
Iridium – Element information, properties and uses | Periodic Table.
Related searches
- what is the atomic mass of cesium?
- how to tell how many neutrons an atom has
- how to know how many neutrons an atom has
- what is the atomic mass of cesium
- how many protons does iron have
- how many atoms are in tungsten
- the atomic number of cesium is 55
- how many electrons are in tungsten
- how many neutrons does tungsten have
- how many neutrons does chlorine have
- tungsten atomic mass
- how many neutrons are in an atom of tungsten
- how many neutrons does sodium have
- how many neutrons in tantalum
- how many neutrons does nickel have
- how to get neutrons of an atom
- how many neutrons are in an atom of titanium
Information related to the topic how many neutrons does an atom of tungsten have
Here are the search results of the thread how many neutrons does an atom of tungsten have from Bing. You can read more if you want.
You have just come across an article on the topic how many neutrons does an atom of tungsten have. If you found this article useful, please share it. Thank you very much.