Let’s discuss the question: how many atoms are there in 15.0 grams of sulfur. We summarize all relevant answers in section Q&A of website Linksofstrathaven.com in category: Blog Finance. See more related questions in the comments below.

How many atoms are in a gram of sulfur?
How many atoms are in 1 gram of sulfur? Explanation: Atoms of sulfur = 9.60⋅g32. 06⋅g⋅mol−1×6.022×1023⋅mol−1 . Because the units all cancel out, the answer is clearly a number, ≅2×1023 as required.
How many atoms are in 16g of sulfur?
We know that S has a molar mass of 32 g/mol . Therefore, there would be 3.01⋅1023 atoms of sulfur in 16 g of pure sulfur powder.
How To Convert Grams to Atoms – THE EASY WAY!
Images related to the topicHow To Convert Grams to Atoms – THE EASY WAY!

How many atoms are there in 16.3 g of sulfur?
Thus, there are 3.06 × 1023 atoms of S in 16.3 g of S.
How many atoms are present in 10.0 grams of sulfur?
6.02 x 10 atoms S O 10.0 g S x 1.0 g S LEGO.
How many atoms are in 32g of sulfur?
Answer and Explanation: 32 grams of sulfur will contain 6.022 X 1023 sulfur atoms. To find this answer, we begin by converting the sample size of 32 grams to moles.
How many atoms are there in sulfur?
| Group | 16 | 115.21°C, 239.38°F, 388.36 K |
|---|---|---|
| Atomic number | 16 | 32.06 |
| State at 20°C | Solid | 32S |
| Electron configuration | [Ne] 3s23p4 | 7704-34-9 |
| ChemSpider ID | 4515054 | ChemSpider is a free chemical structure database |
How many atoms are there in 40g of sulphur?
Expert-verified answer
The number of atoms present in 40g of Sulphur is 7.527 × 10²³ atoms. One mole of any substance is equal to 6.022 ×10²³ which is called Avogadro’s constant. Number of atoms present in 40g of Sulphur is 7.527 × 10²³ atoms.
How many molecules are in 64 grams of sulphur?
∴ 64 g of sulphur will contain=6.022×1023×64256=1.506×1023 molecules.
How many grams are in sulfur?
One atom of sulfur has a mass of 32.07 amu; one mole of S atoms has a mass of 32.07 g. For compounds, the molecular mass (in amu) is numerically the same as the mass of one mole of the compound in grams.
How do you calculate grams to atoms?
To calculate the number of atoms in a sample, divide its weight in grams by the amu atomic mass from the periodic table, then multiply the result by Avogadro’s number: 6.02 x 10^23.
How much atoms are in a mole?
The value of the mole is equal to the number of atoms in exactly 12 grams of pure carbon-12. 12.00 g C-12 = 1 mol C-12 atoms = 6.022 × 1023 atoms • The number of particles in 1 mole is called Avogadro’s Number (6.0221421 x 1023).
Calculate the Mass of a Single Atom or Molecule
Images related to the topicCalculate the Mass of a Single Atom or Molecule
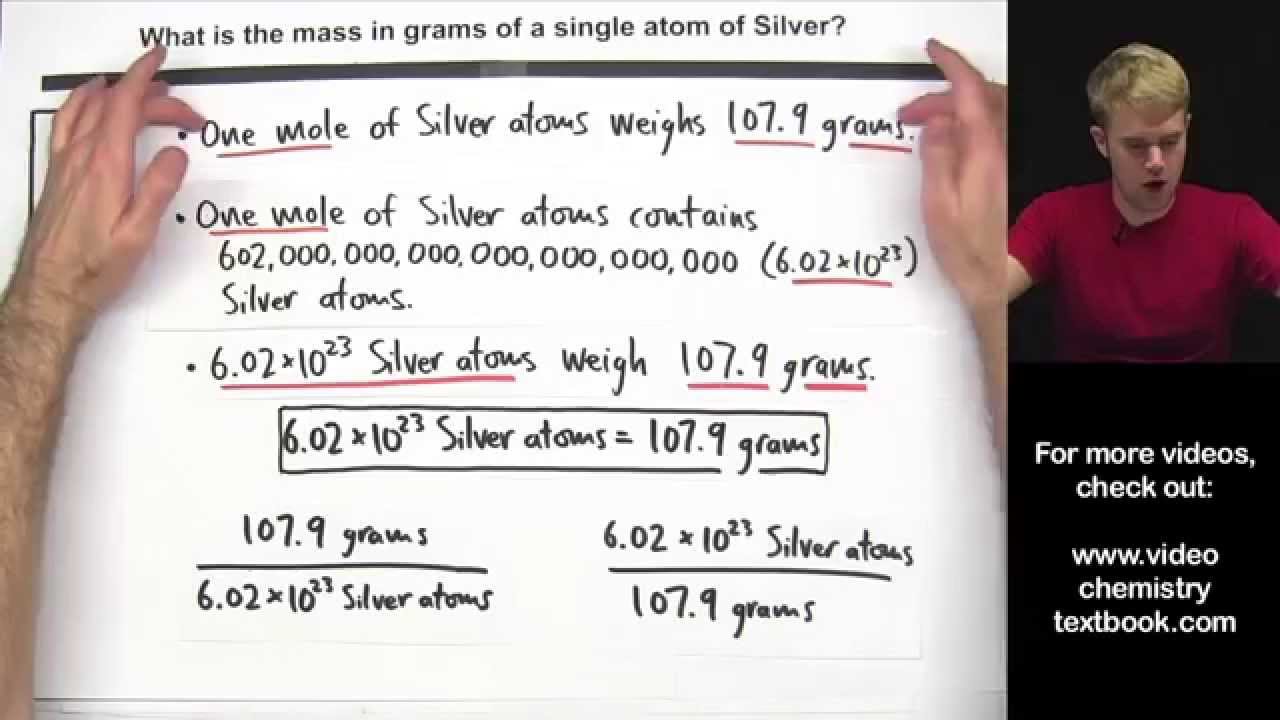
What is the mass number of sulfur?
How many atoms are in 8 grams of sulphur?
Your question, though, was how many atoms, so you have to multiply that number which is how many molecules by 8 atoms per molecule of sulphur. So you get 1.503 x 10^23 atoms in 8 grams of sulfur.
How many atoms are in 16 g of oxygen and 8 grams of sulphur?
Answer. therefore we hai 6.022×10^23 atoms.
How many atoms are in 4 moles Al?
Therefore, \[2.41\times 10{}^\text{2}\] atoms/mol of Aluminum are present in $4$ mol of Aluminum.
How many atoms does 32g of sulfur contain Brainly?
Answer. Therefore 32 GMs of sulfur consists of 6.022 x 10^23 atoms.
How many moles are in 32g of sulfur?
So 32 g of sulphur is 1 mole of sulphur.
How many atoms are there in 32g of oxygen?
One mole of oxygen gas, which has the formula O2, has a mass of 32 g and contains 6.02 X 1023 molecules of oxygen but 12.04 X 1023 (2 X 6.02 X 1023) atoms, because each molecule of oxygen contains two oxygen atoms.
What type of atoms are in sulfur?
It is classified as a nonmetal. Sulfur atoms have 16 electrons and 16 protons with 6 valence electrons in the outer shell. Sulfur is the tenth most abundant element in the universe. Sulfur can take the form of over 30 different allotropes (crystal structures).
How many gram atoms are present in 640 gram of sulphur?
∴ 640 g of sulphur =132×640=20g atoms of sulphur.
Chemistry – How many carbon atoms are there in 200.0 g of carbon dioxide?
Images related to the topicChemistry – How many carbon atoms are there in 200.0 g of carbon dioxide?

How many atoms are in 25.6 grams of sulphur molecules?
There are 2.37×1023 2.37 × 10 23 sulfur atoms present in 25.6 g of Al2(S2O3)3 A l 2 ( S 2 O 3 ) 3 .
How many moles of H atoms are there in 0.5 mole of H2 S o4?
So in 0.5 moles, there are 3 x 10^23 molecules. There are 4 H atoms per molecule. Therefore, 0.5 moles contains 12 x 10^23 atoms of H.
Related searches
- how many grams are in 9 03 x 1023 atoms of copper cu
- how many atoms are in 3.5 moles of arsenic?
- how many moles are in 2.70 x 10 24 atoms of copper cu
- how many atoms of boron are present in 8.0 x 10 6 moles of boron
- how many atoms of sulfur
- how many atoms of boron are present in 8 0 x 10 6 moles of boron
- given 88 0 grams of boron how many atoms of boron b are present
- how many atoms are in 15 grams of sulfur
- how many atoms are in 1 gram of sulfur
- how many atoms are in 4 3 grams of solid sulfur
- how many atoms are there in 15.0 grams of sulfur (s)
- how many atoms are in sulfur
- how many moles are in 2 70 x 10 24 atoms of copper cu
- how many atoms are in 3 5 moles of arsenic
- how many atoms are in 4.3 grams of solid sulfur?
Information related to the topic how many atoms are there in 15.0 grams of sulfur
Here are the search results of the thread how many atoms are there in 15.0 grams of sulfur from Bing. You can read more if you want.
You have just come across an article on the topic how many atoms are there in 15.0 grams of sulfur. If you found this article useful, please share it. Thank you very much.
