Let’s discuss the question: how metals displace hydrogen from water. We summarize all relevant answers in section Q&A of website Linksofstrathaven.com in category: Blog Finance. See more related questions in the comments below.
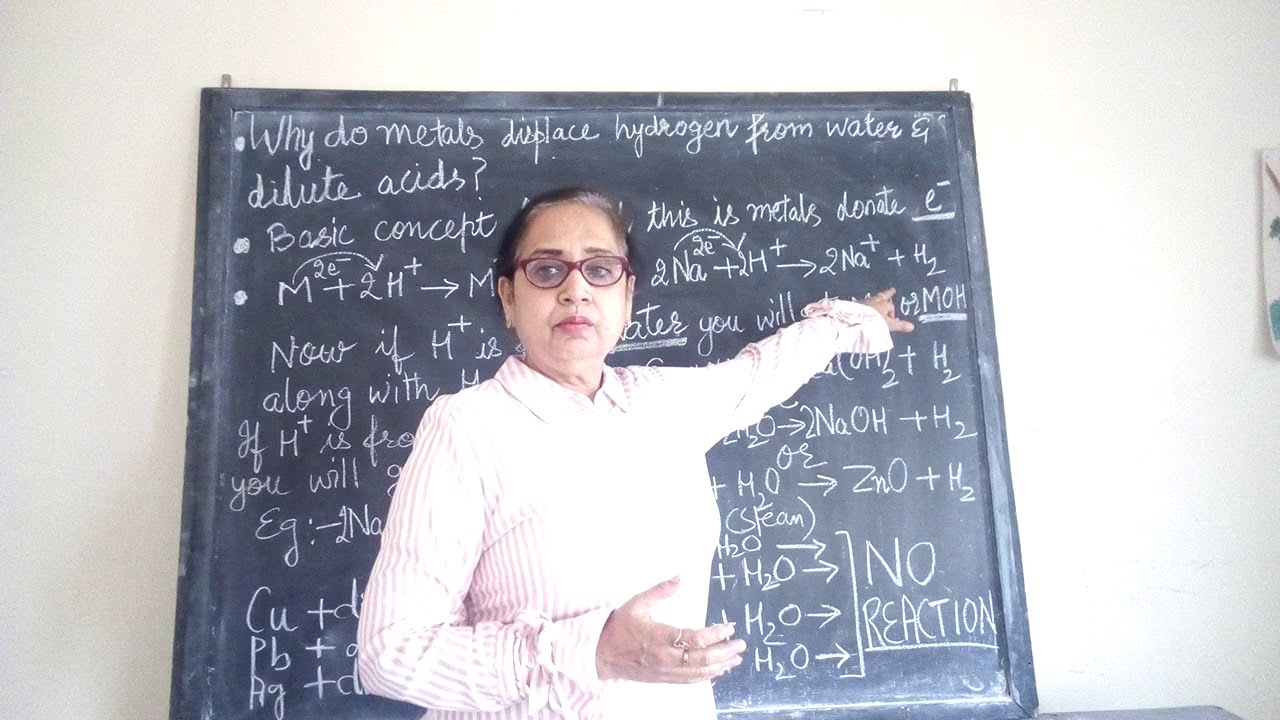
How metals displace hydrogen from water explain?
Sodium and potassium react violently with cold water to an extent that hydrogen gas so produced catches fire. 2. Calcium reacts less violently with water.Ca + 2H2O → CaOH2 + H2 ↑3. Magnesium reacts with hot water to form magnesium oxide and hydrogen.Mg + H2O → MgO + H2 ↑4.
How do metals displace hydrogen from dilute acids?
The series of metals in decreasing order of reactivity is called reactivity or activity series of metals. The metals at the top are most reactive whereas metals at the bottom are less reactive. The metals above hydrogen are more reactive than hydrogen. They can displace hydrogen from dilute acids and water.
Why do metals displace Hydrogen from dilute acids and water?
Images related to the topicWhy do metals displace Hydrogen from dilute acids and water?
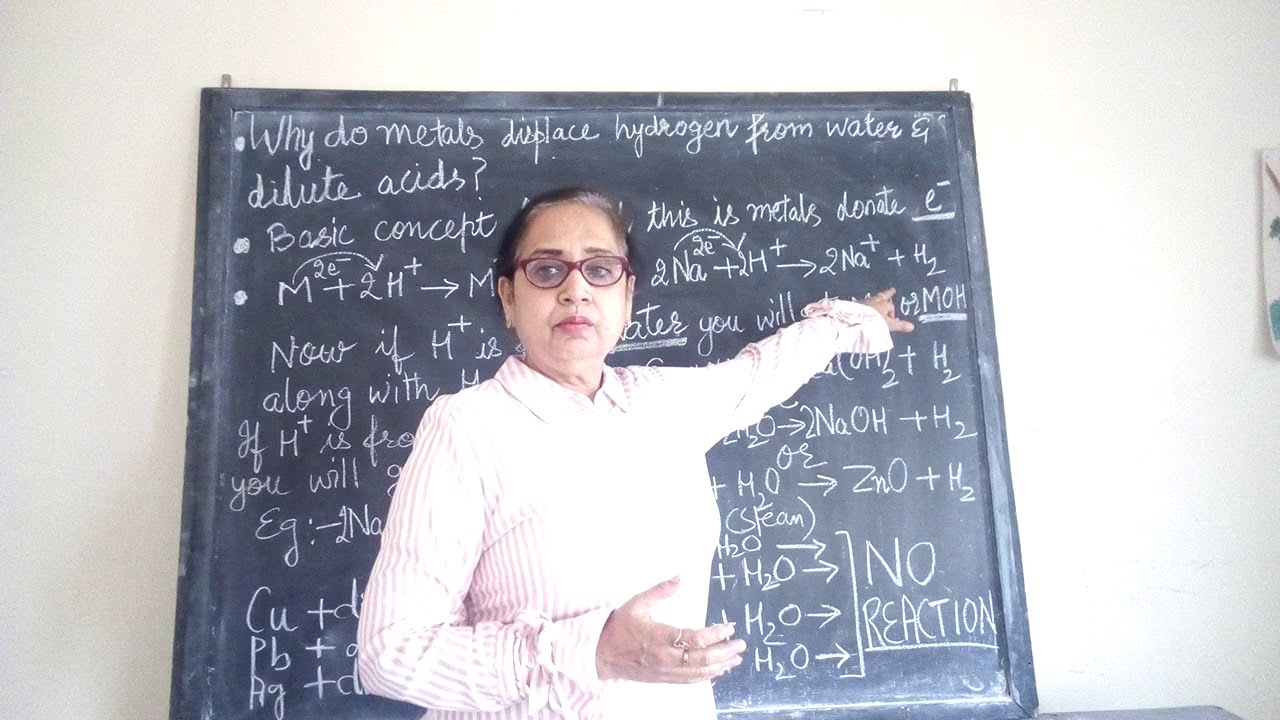
Why do metals displace hydrogen?
Metals displace hydrogen from acids because they are above hydrogen in the reactivity series, resulting in the formation of a salt and hydrogen gas.
Where do metals which can displace hydrogen from water at room?
Calcium is very reactive metal so it displaces hydrogen from water at room temperature and form a strong base calcium hydroxide.
How do you displace hydrogen?
Hydrogen is displaced by the metals from acids that are placed above hydrogen in the reactivity series of the metals. This is because these metals are more reactive than hydrogen. Reaction of calcium with hydrochloric acid. Here , Hydrogen is displaced by Calcium from Hydrochloric acid .
Which metal can displace hydrogen from water or acids?
Copper – Gold are below Hydrogen and lean to the least reactive end. All of the elements above Hydrogen in the reactivity series will displace Hydrogen atoms from an acid during a reaction.
What displaces hydrogen from boiling water?
Calcium reacts with cold water to form calcium hydroxide with the liberation of hydrogen gas.
Which elements can displace hydrogen from dilute acids?
So, aluminium, and magnesium, or any other metal placed above hydrogen can be considered as an example of the metals that displace hydrogen from dilute acids; and mercury, and silver are the metals which will not displace hydrogen.
Do all metals displace hydrogen when react with dilute acids give reason?
Solution : In displacement reaction, more reactive metal displaces less reactive metal. Since, zinc is more reactive than hydrogen it displaces hydrogen in dilute hydrochloric acid and copper is less reactive than hydrogen and hence cannot displace hydrogen in dilute hydrochloric acid.
What does displacement of hydrogen mean?
An explosion occurs because when hydrogen is displaced from water, it quickly forms hydrogen gas. The reaction is also exothermic (meaning it releases heat) which causes the hydrogen gas to ignite.
What reacts with water to form hydrogen?
Most alkali metals and alkaline earth metals react with water to produce hydrogen. The alkali metals comprise Group 1 of the periodic table, and include lithium, sodium, potassium, rubidium, cesium and francium.
Metals can displace hydrogen from water|Chemistry|ClassX| Digital Generation
Images related to the topicMetals can displace hydrogen from water|Chemistry|ClassX| Digital Generation

How do different metals react with water?
Metals react with water and produce a metal oxide and hydrogen gas. Metal oxides that are soluble in water dissolve in it to further form metal hydroxide. But all metals do not react with water. Metals like potassium and sodium react violently with cold water.
Which of the following metal will displace hydrogen from cold water?
If we look at the activity series, only sodium and calcium can displace hydrogen from cold water. Silver, and chromium, are being less active, hence they cannot displace the hydrogen from cold water.
Does silver displace hydrogen?
Because silver is less reactive than hydrogen. Since it is less reactive it cannot displace hydrogen from dilute acid solution.
Which metals can displace hydrogen during a single displacement reaction?
Based on the activity series of metals, calcium can displace hydrogen from water, but iron cannot displace chromium from chromium(II) sulfate.
Which metal would readily displace hydrogen from steam?
Explanation. In the electrochemical series, H(^+) is above all the other elements in the series except Magnesium. Hence, only Mg(^{2+}) can displace hydrogen from a reaction.
Can lithium displace hydrogen from water?
…
The Activity Series.
| Most Active (Easily Oxidized—Readily Lose Electrons) | ||
|---|---|---|
| lithium | Li | These metals displace hydrogen from water Ca(s) + 2H2O(l)→ Ca(OH)2 + H2 (g). These elements are very reactive and react readily to form compounds. |
| potassium | K | |
| barium | Ba | |
| calcium | Ca |
Which of the following metals Cannot replace hydrogen from water?
Option D – Copper
Metals above hydrogen in the activity series can displace or replace hydrogen present in water (H2O) example of such metals are Ca, K, Na etc. But the metals below hydrogen in the activity series cannot replace hydrogen in the activity series example of such metals are Cu, Hg.
What metals can replace hydrogen in HCL?
The six metals from iron to lead will replace hydrogen from hydrochloric, sulfuric and nitric acids. Their oxides can be reduced by heating with hydrogen gas, carbon, and carbon monoxide. All the metals from lithium to copper will combine readily with oxygen to form their oxides.
Which element is most likely to replace hydrogen and displacement reaction?
For example, lithium will react with cold water, replacing hydrogen. It will also react with steam and with acids, since that requires a lower degree of reactivity.
Does Aluminium displace hydrogen?
Aluminium displaces hydrogen from dilute HCl whereas silver does not. The E.M.F. of the cell prepared by combining A/Al3+ and Ag/Ag+ is 2⋅46V.
Collection of Hydrogen by Water Displacement
Images related to the topicCollection of Hydrogen by Water Displacement

How many metals in the following list can displace hydrogen from boiling water?
Thus, the list shows 14 metals that will displace hydrogen from acids.
Which of these Cannot displace hydrogen from a dilute acid?
Silver cannot displace hydrogen from a dilute acid because it is a less reactive noble metal and does not react with acids.
Related searches
- how many metals in the following list can displace hydrogen from boiling water
- can magnesium displace hydrogen from water
- can copper displace hydrogen from water
- explain how metals displace hydrogen from water
- can non metals displace hydrogen from water
- which metal can displace hydrogen from water
- can zinc displace hydrogen from water
- reactivity series
- which metals displace hydrogen from water
Information related to the topic how metals displace hydrogen from water
Here are the search results of the thread how metals displace hydrogen from water from Bing. You can read more if you want.
You have just come across an article on the topic how metals displace hydrogen from water. If you found this article useful, please share it. Thank you very much.